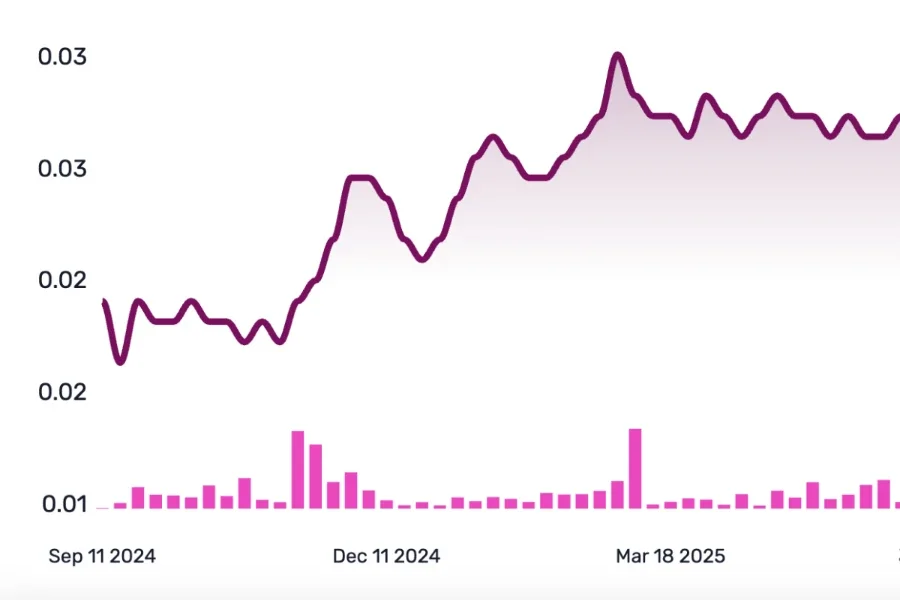
News and
Announcements
Stay up to date on all our news and market announcements.
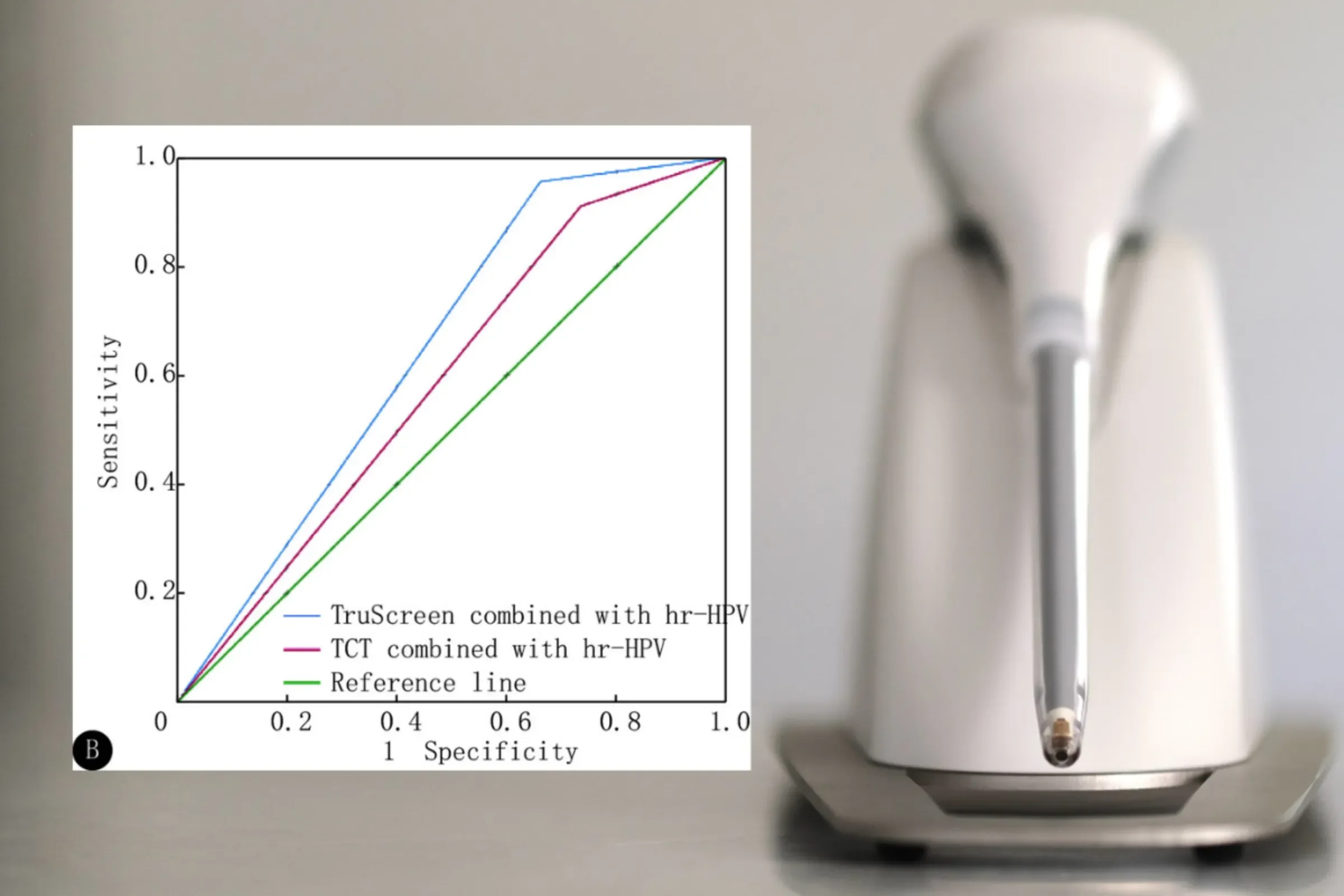
Sichuan University clinical study confirms superiorityof TruScreen + hr-HPV co-testing
An independent clinical study comparing TruScreen with Thinprep cytology test (TCT), and in combination with high-risk human papillomavirus (hr-HPV) testing, has been published and peer-reviewed in Journal of Sichuan University, confirming superiority of TruScreen + hr-HPV co-testing.
Spark+ Research Flash Note
Spark+ Investor Relations Research has published a Research Flash Note on TruScreen Group Limited.
TruScreen validated as Superior Primary Screening Tool by World's largest Opto-Electronic Cervical Cancer Screening Study
Results of the world's largest ever (n=14,982) study of opto-electronic cervical cancer screening, conducted by Chinese Obstetricians and Gynaecologists Association (COGA), have been published validating TruScreen as a superior primary cervical cancer screening tool over Liquid Based Cytology (LBC) and high-risk Human Papillomavirus (hrHPV) testing.
Key Markets Update - Feb 2026
TruScreen Group Limited is pleased to provide an update on activities in key markets: Uzbekistan, Zimbabwe, Central Asia, Europe and China.
Early Warning Signs of Cervical Cancer
Recognizing the early warning signs of cervical cancer is critical for timely diagnosis and treatment. Although early stage cervical cancer frequently occurs without symptoms, awareness of the signs can help enable early detection and treatment to improve outcomes.
The Rising Risk of Cervical Cancer in Women Over 65: A Call for Continued Screening and an Effective Screening Solution
There is a significant global gap in cervical cancer prevention for older women. Reports from public health platforms in China and other parts of Asia reveal that large numbers of older women, especially those in rural or low-resource settings, have never participated in cervical screening programs. This gap places them at high risk for undiagnosed and advanced-stage cervical cancer, with limited treatment options and worse outcomes.
Clinical study validates TruScreen’s efficacy and safety for pregnant women
Leading Chinese medical publication Family Doctor confirms TruScreen’s efficacy as a cervical cancer screening tool for pregnant women, in a 2,000-patient study conducted at Guilin People’s Hospital.
TruScreen investigates Commercial Opportunities for Western Europe and Middle East
TruScreen has authorised it's Indian distributor, Renovate Biologicals Pvt Ltd, to investigate commercial and market opportunities in the Middle East and Western Europe from 1 January 2026.
In support of World Cervical Cancer Elimination Day
This year’s World Cervical Cancer Elimination Day theme “Act Now: Eliminate Cervical Cancer” calls for bold, united action to build on existing progress and accelerate impact toward the World Health Organization’s 90-70-90 targets by 2030. Read how TruScreen’s innovative, real-time, AI-enabled technology makes cervical screening more accessible, accurate, and sustainable - to support increased screening rates globally.
TruScreen appoints distributor to South Africa
TruScreen has appointed Johannesburg distributor AIR to distribute its AI enabled TruScreen cervical cancer screening system in South Africa. This is a key market for TruScreen’s African strategy that will grow TruScreen’s African distribution reach to 30 million women of screening age.
Martin Dillon, CEO, presents at Aussie Equities Day, 3 Oct 2025, Singapore
TruScreen is delighted to be presenting at Aussie Equities Day 2025 hosted by Spark Plus on Friday 3 Oct, during Singapore's F1 GP Week.
Misread smear leads to cervical cancer diagnosis delay, $18,000 fertility bill
A young Auckland real estate agent is battling stage 3 cervical cancer after a misread smear test delayed her diagnosis by a year. Such stories highlight the inherent risk of using ageing methodologies with subjective human interpretation to screen for cervical cancer.
Annual Shareholders Meeting Presentation
View TruScreen’s Annual Shareholder Meeting presentation, delivered today by Martin Dillon, CEO.
Public Screening Programs in Uzbekistan and Northeast India
An update on the implementation of our strategy to engage with Ministries of Health, Non-Government Organizations (NGO’s) and private foundations funding public screening programs in emerging markets.
SPP Closes Oversubscribed
The Share Purchase Plan announced on 29 May 2025 closed oversubscribed raising ~NZ$1.67 million (~83 million shares at NZ$0.02).
Key Markets Update
Uzbekistan regulatory approval received; Zimbabwe validation to be completed 21 July; First TruScreen order from India landed; TruScreen’s Superiority Confirmed in Beijing Study.
TruScreen Raises $2.3 Million in Share Placement
Truscreen is pleased to advise that the Company has received firm commitments from both new and existing investors for an initial Placement of 107,034,091 shares at NZ$0.022/A$0.02 each to raise NZ$2,354,750, before costs.
TruScreen announces the opening of a NZ$3m Capital Raise
Following a year of significant progress growing our geographic distribution capability (India and Indonesia, among others) and product suite (Dalton Bio HPV IVD products), TruScreen is seeking approximately NZ$3m through Placement, Share Purchase Plan and Options to fund further growth.
TruScreen appointed to distribute DaltonBio’s HPV detection products in India
TruScreen is pleased to announce that an Agreement has been signed with Hangzhou Dalton Bioscience Limited (“DaltonBio”), for TruScreen to distribute DaltonBio’s HPV-related In Vitro Diagnostics (“IVD”) products in India.
Sign up for regular
updates and
announcements.
Can’t see the form?
Email us at marketing@truscreen.com to subscribe